Les maladies gastro-intestinales, qui vont des problèmes fonctionnels aux cancers, font partie des affections les plus courantes dans le monde. À lui seul, le reflux gastro-œsophagien (RGO) touche environ 1,2 milliard de personnes dans le monde, et sa prévalence augmente dans toutes les régions du globe. De nombreuses maladies gastro-intestinales sont difficiles à diagnostiquer car les symptômes sont souvent vagues et variables. En outre, les diagnostics actuellement disponibles nécessitent une interprétation hautement qualifiée et un coût d’installation élevé, avec un manque d’accès considérable. Par exemple, le RGO fait l’objet d’un test de diagnostic définitif dans moins de 2 % des cas.
Diagnostic des maladies gastro-intestinales grâce à eCAP
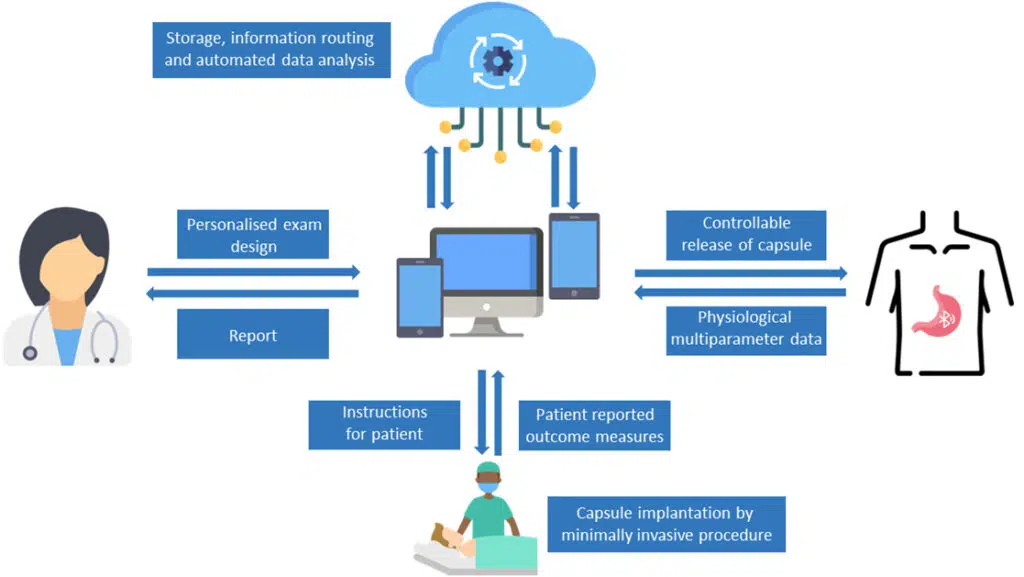
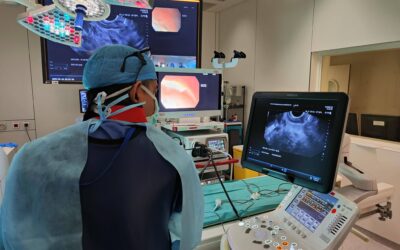
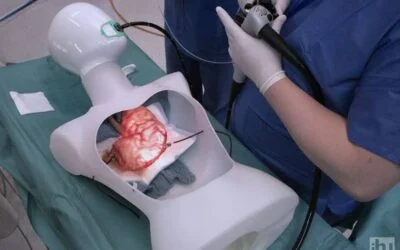
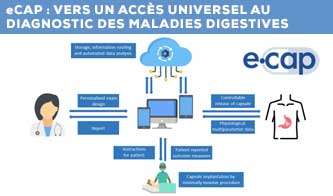
Le consortium eCAP développera une capsule intelligente multi-détection, implantée par des techniques mini-invasives, qui permet de surveiller la physiologie gastro-intestinale pendant une période contrôlée. Grâce aux normes de communication des smartphones et à l’informatique en cloud, des données en temps réel sur l’expérience des patients seront collectées au moyen d’une application interactive conviviale. La gestion de la maladie par le patient sera soutenue par l’interprétation précise des données par les médecins, aidés par l’intelligence artificielle. « Un test de dépistage du reflux acide plus précis, plus rentable et plus convivial pour le patient est aujourd’hui un besoin de plus en plus urgent auquel les solutions actuelles sur le marché ne répondent pas. Ce qui est passionnant, c’est la réunion de ce groupe particulier de partenaires, particulièrement apte à résoudre le problème des maladies gastro-intestinales qui touchent les gens dans le monde entier« , explique Lee Swanström, coordinateur du projet à l’IHU de Strasbourg. « Lorsque nous avons entamé les discussions, nous avons eu d’excellentes idées pour étendre la preuve de concept initiale, même au-delà du dépistage du RGO, de sorte que le projet a un énorme potentiel. Nous remercions vivement la communauté européenne d’avoir reconnu notre vision en soutenant ce projet. »
eCAP se déroulera sur quatre ans et s’appuiera sur une expertise de niveau mondial. Le projet est dirigé par l’IHU de Strasbourg (FR). Les solutions techniques seront mises au point par le Tyndall National Institute (IE), Sentron Europe BV (NL), Integer (IE) et Enterasense (IE), sur la base de la conception utilisateur d’Imagination Factory (UK). Les études cliniques au Kenya et en Ukraine seront menées par l’Association des praticiens des blocs opératoires du Kenya (OTPAK, KE) et par une clinique de gastroentérologie et d’endoscopie de Kiev – Olymed (UA), qui continue de participer activement au projet, malgré les évènements dévastateurs qu’ils subissent aujourd’hui. L’analyse économique de la santé sera réalisée par Betthera (CZ) et la gestion du projet et les activités de diffusion par AMIRES, The Business Innovation Management Institute (CZ).
Consortium du projet :
Durée du projet : 1er mai 2022 – 30 avril 2026
Contacts presse :
– Lee Swanström (Coordinateur du projet) lee.swanstrom@ihu-strasbourg.eu
– Kristin Saar (Chef de projet) saar@amires.eu